Don’t want to miss the latest professional materials and information!
You can click on the “Starch and Starch Sugar Handbook” above – top right “…” to bookmark
Honey

Starch sugar is a type of sugar produced from starchy foods such as grains and tubers using acid, acid-enzyme, or full enzyme methods, including maltose, isomaltose, glucose, fructose, high fructose syrup, sorbitol, and various oligosaccharides, collectively referred to as starch sugar. Starch sugar has a long history in China, with detailed methods of sugar production from rice described in the “Essential Techniques for the People” over 1600 years ago.
As the production of starch sugar in China has increased year by year, and the price of white sugar has risen, beekeepers often use starch sugar as feed for bees, sometimes intentionally or unintentionally mixing it into the harvested commercial honey.
High fructose syrup is visually and compositionally similar to honey, making it a preferred raw material for producing fake honey and adulterating honey.
Stable carbon isotope ratio analysis and ion chromatography are effective methods for identifying starch sugar adulteration in honey. The testing methods are complex and costly, necessitating research to propose simpler methods suitable for honey purchasing and operating units.
There are many varieties of starch sugar; here we will briefly introduce maltose and high fructose syrup that may be used in honey.
Maltose
Maltose is a disaccharide formed by the condensation of two glucose molecules, produced from starch using acid or starch enzymes from malt or bran. It has a high maltose content and low glucose content, also known as syrup sugar, with a sweetness equivalent to 50% of the same concentration of sucrose solution. There are several methods for producing maltose. Currently, the production process involves liquefying starch with α-amylase solution, followed by saccharification with β-amylase or α-fungal amylase, and then filtered and decolorized 1-2 times for refinement.
High Fructose Syrup
High fructose syrup is produced from starch as raw material, liquefied by starch enzymes, saccharified by glucose amylase, and then decolorized, filtered, and refined through ion exchange, resulting in F42 high fructose syrup containing more than 42% fructose.
F42 high fructose syrup generally contains more than 42% fructose and 50% glucose, and is prone to crystallization at lower temperatures, while F90 ultra-high fructose syrup, with over 90% fructose in its sugar composition, has a sweetness equivalent to 110% of the same concentration of sucrose solution. F90 ultra-high fructose syrup can be blended with F42 high fructose syrup to produce F55 high fructose syrup with over 55% fructose content, which is very popular in the food, brewing, and pharmaceutical industries.
In the 1940s, with the development of industrial enzyme preparations, significant progress was made in starch sugar production technology.
In the early 1960s, pure crystalline glucose was produced using enzyme preparations and new processes.
By the late 1960s, isomerase was developed to convert glucose into fructose.
In the 1970s, the United States and Japan began using dual enzyme methods to produce high fructose corn syrup.
High fructose syrup is colorless and odorless, flows well at room temperature, is easy to use, and is inexpensive. It can partially or completely replace sucrose in beverage production and food processing, and has a rich flavor that helps maintain the original fruit aroma in fruit juice beverages.
China introduced the dual enzyme method for producing high fructose syrup in the 1980s, and due to the research and development of enzyme preparations and production processes, there are now more than 30 varieties of starch sugar. Most high fructose syrup is made from corn starch, also known as high fructose corn syrup; others are made from broken rice, sweet potatoes, and potatoes.
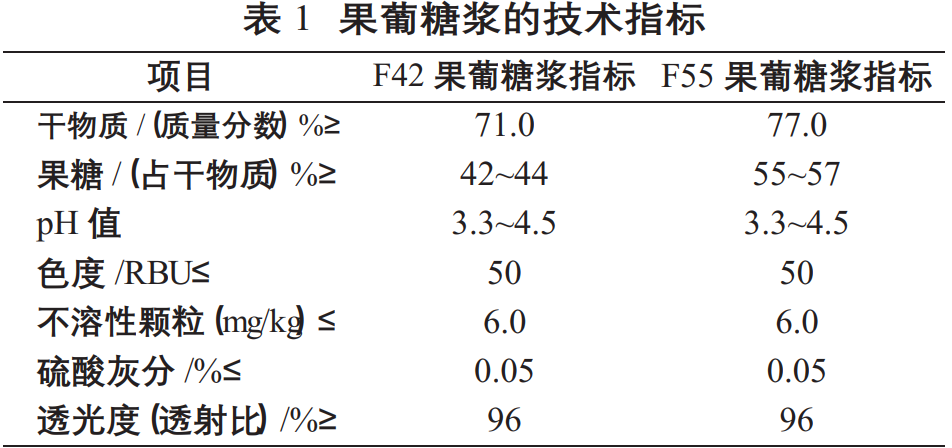
Technical Indicators of High Fructose Syrup:
High fructose syrup is a colorless or light yellow, clear, transparent, viscous liquid; it has a unique aroma of high fructose syrup; its sweetness is mild and free of off-flavors.
The technical indicators of high fructose syrup produced by sugar factories are shown in Table 1 and must comply with food hygiene standards.
For Bee Feed:
Maltose syrup and high fructose syrup can replace white sugar for bee feed. It is important not to use them during the honey collection period to avoid mixing them into commercial honey.
Maltose and F42 high fructose syrup are prone to crystallization at lower temperatures, suitable for use in spring and summer, while F55 high fructose syrup can be used as winter feed.
It is essential to purchase high-quality, high-concentration high fructose syrup, as those with high moisture content can ferment easily in high temperatures. Additionally, high fructose syrup should not be stored at high temperatures for long periods, as some fructose can convert into harmful hydroxymethylfurfural for bees.

Stable Carbon Isotope Ratio Analysis Method (SCIRA)
The stable carbon isotope ratio refers to the ratio of two carbon isotopes (such as 12C and 13C) in a substance, abbreviated as 13C value or ratio.
The raw material for producing high fructose syrup, corn, is a C4 plant, with a 13C value of -20‰~-10‰; almost all honey source plants are C3 plants, with a 13C value range of -28‰~-22‰.
While the carbohydrates (sugars) formed by these two types of plants are chemically identical, they differ in their carbon isotope ratios.
If these two types of carbohydrates are mixed, the carbon isotope ratio of the mixture will change based on the mixing proportions.
This principle underlies the use of stable carbon isotope ratio analysis to identify the adulteration of honey with high fructose syrup.
Through the analysis of test results from mixing high fructose syrup into honey in varying proportions, the conclusion is that a ratio less than -23.5‰ indicates pure honey; a ratio greater than -21.5‰ indicates honey adulterated with high fructose syrup. Samples with ratios between -23.5‰ and -21.5‰ fall into an indeterminate “gray zone.”
Research has shown that using the proteins in honey as internal standards can effectively identify adulterated honey in the gray zone, establishing an internal standard carbon isotope testing method for honey.
This method calculates the difference between the 13C value of honey protein and the 13C value of honey, referred to as the ISCIRA index.
An ISCIRA index of -1‰ indicates that the honey contains 7% high fructose syrup. The smaller the index, the more high fructose syrup is present.
Ion Chromatography Method
The national standard for the determination of starch sugar adulteration in honey – ion chromatography method (GB/T21533-2008) specifies the determination methods for high fructose syrup, maltose syrup, isomaltose syrup, and syrup sugars in honey.
This standard applies to the determination of starch syrup in honey, with a detection limit of 5% starch syrup. The detection effect is good for starch sugars made from C3 plants such as broken rice, sweet potatoes, and potatoes.
Detection Principle:
Honey contains almost no oligosaccharides with a degree of polymerization (DP) greater than 5, while various starch syrups contain oligosaccharides with DP greater than 5. Using gel permeation chromatography to remove fructose and glucose from the sample, the oligosaccharides are enriched and then directly detected using anion exchange chromatography-electrochemical detector, with the presence of oligosaccharides with DP greater than 5 serving as the criterion for determining starch syrup in honey.
Result Determination:
By analyzing and comparing the oligosaccharide profiles of pure honey negative control samples and honey positive control samples containing 5% syrup, distinct differences in the “fingerprint area” can be identified, which serves as an indicator for determining starch syrup adulteration in pure honey.
Any honey sample adulterated with high fructose syrup will exhibit two typical “fingerprint peaks” P1 and P2 between maltotriose and maltotetraose, and between maltotetraose and maltopentaose, indicating the presence of high fructose syrup in honey.
Any honey sample adulterated with maltose syrup will show three typical “fingerprint peaks” P1, P2, and P3 between maltotriose and maltotetraose, maltotetraose and maltopentaose, and after maltopentaose, indicating the presence of maltose syrup (including high maltose syrup, isomaltose syrup, and syrup sugars).
In addition to the basic characteristics described, other peaks with different peak shape characteristics can be observed under different production conditions, but they do not affect the fundamental characteristics and determinations of the “fingerprint area.”
The national standard appendix includes positioning chromatograms of maltose standard mixed solutions, as well as oligosaccharide chromatograms of pure acacia honey, jujube flower honey, linden tree honey, jujube honey, and rapeseed honey; as well as oligosaccharide chromatograms of different honey types mixed with 5% different high fructose syrup and maltose syrup as references.
Microscopic Inspection Method
Kerkvliet et al. proposed a method for detecting sugar and sugar product adulteration in honey using microscopy, based on the principle that sucrose contains many thin-walled cells, single rings of circular vessels, and epidermal cells from sugarcane. By examining honey samples with polarized light microscopy, orthogonal polarization, and first-level red blocking plates, it was found that honey adulterated with sucrose would reveal sugarcane cells.
Subsequent studies indicated that if more than 150 thin-walled cells and hardened cells, or 10 rings, are detected in a 10g honey sample, it indicates that the honey is adulterated with more than 7% sucrose.
The microscopic inspection method warrants further research, as the required instruments and operations are relatively simple and suitable for the needs of honey purchasing and operating units.
It is known that among more than 20 types of bulk honey, each type has its specific pollen (morphological characteristics and size can be found in relevant literature) and pollen spectrum, while starch sugars do not contain pollen.
Research to establish the specific pollen and total pollen unit quantities for each type of bulk honey can serve as a basis for comparison. (Professional sharing; if there are any inaccuracies, please contact for removal)


References:
[1] WHITE J W, DONER L W, The 13C/12C ratio in honey [J]. J Apic Res, 1978, 17:94-99.
[2] PAN Jianguo, DENG Zhaohua, Latest Developments and Applications of Stable Carbon Isotope Ratio Analysis Method (SCIRA), Chinese Beekeeping, 2002, (1): 9-10, (2): 11, 16.
[3] WHITE J W, WINTERS K, Honey protein as an internal standard for stable isotope ratio detection of adult adulteration of honey [J]. J Assoc Off Anal Chem, 1989, 81:610-619.
[4] KERKVLIET J D, SHRESTHA M, TULADHAR K, Manandhar, Microscopic detection of adult adulteration of honey with cane sugar and cane sugar products [J]. Apidologie, 1995, 26:131-139.
[5] KERKVLIET J D, HARRO A J MEIJER, Adult adulteration of honey: relation between microscopic analysis and C measurements [J]. Apidologie, 2000, 31:717-726.
Facing the Sun, Striving Every Day


