Editor’s Note
Glucagon-like peptide-1 receptor agonists (GLP-1RA) have been used for nearly 20 years in the management of blood glucose in patients with type 2 diabetes (T2DM) since their launch. With the increasing evidence from research, the efficacy of some GLP-1RAs in effective glucose lowering, multiple metabolic improvements, and cardiovascular benefits has been widely recognized. Their recommended status in authoritative guidelines both domestically and internationally has been continuously elevated, making them a priority recommendation in multiple clinical application pathways, benefiting an increasingly broad patient population. Semaglutide tablets, as the only oral GLP-1RA currently available globally, have promoted the early and widespread application of GLP-1RAs in T2DM populations. Meanwhile, the drug safety and patient tolerance of GLP-1RAs have also attracted the attention of many clinical doctors and patients. So, how safe are semaglutide tablets, which will soon be launched in China? This article invites Professor Chen Lulu from Huazhong University of Science and Technology Tongji Medical College Union Hospital to provide a detailed interpretation of the safety data from the phase 3 clinical studies of semaglutide tablets—based on the safety data summary analysis from the PIONEER series studies.
1
Introduction to Semaglutide Tablets
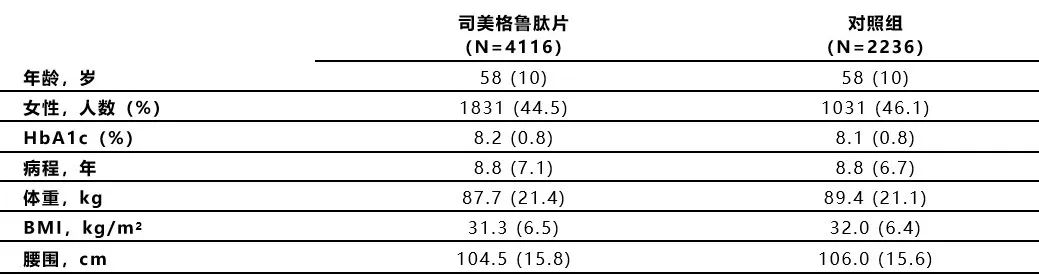
2
Overall Tolerance of Patients is Good,
Gastrointestinal Adverse Events are Most Common
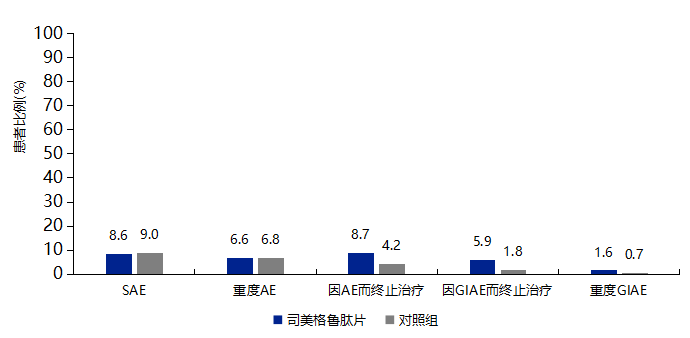
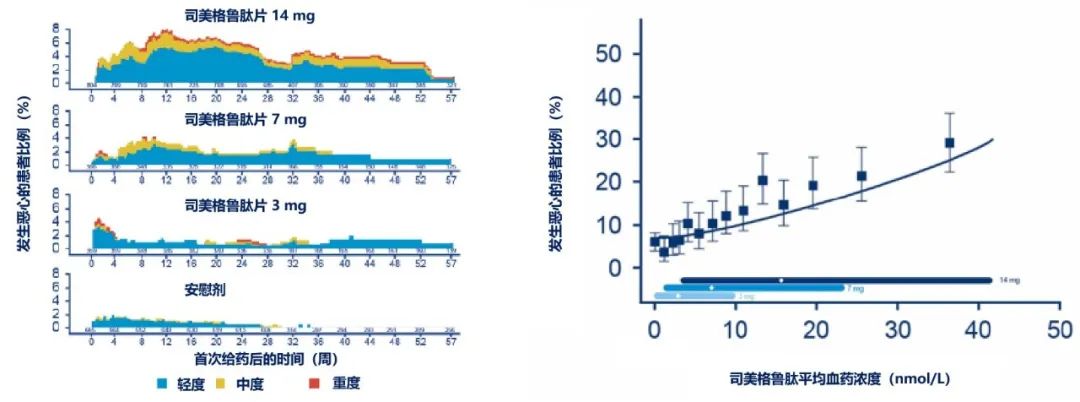
Figure 2 Incidence of Nausea Among Patients in the PIONEER Series
Note: The left figure shows data that only includes placebo-controlled trials in the PIONEER series (PIONEER 1, 4, 5, 7) and demonstrates the severity and patient proportion of nausea among different dose groups of semaglutide tablets and the placebo group over time; the right figure includes data from PIONEER 1-3, 5, 8, 9, showing the distribution range of drug concentrations for different doses of semaglutide tablets, and the relationship between the proportion of patients experiencing nausea and drug concentration.
-
Inform patients that gastrointestinal reactions are the most common adverse reactions, such as nausea and diarrhea. -
Help patients distinguish between increased satiety (one of the drug’s effects) and feelings of discomfort/nausea. -
Most gastrointestinal reactions are mild to moderate and rarely lead to treatment discontinuation. -
Gastrointestinal reactions usually occur early in treatment and tend to lessen over time. -
Start with a small dose and gradually increase to reduce gastrointestinal reactions. -
Avoid discontinuation of treatment as much as possible, based on what the patient can tolerate.
3
Key Areas of Focus
3.1 Semaglutide Tablets Are Safe for Glycemic Control with Low Risk of Hypoglycemia
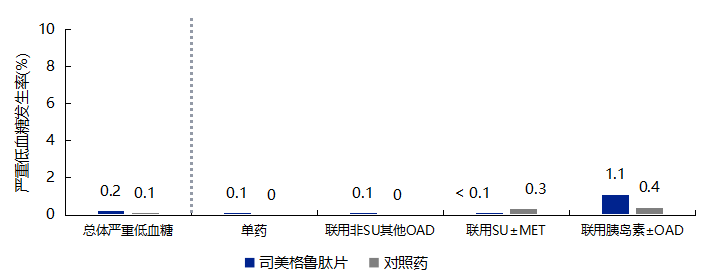
-
Self-monitoring of blood glucose is not required during dose adjustments of semaglutide tablets. -
When semaglutide tablets are used in combination with sulfonylureas or insulin, consideration should be given to reducing the dose of sulfonylureas or insulin to lower the risk of hypoglycemia. -
Self-monitoring of blood glucose is necessary when adjusting doses of sulfonylureas and insulin, especially when initiating semaglutide tablets and reducing insulin dosage.
3.2 No Increased Risk of Malignant Tumors with Semaglutide Tablets
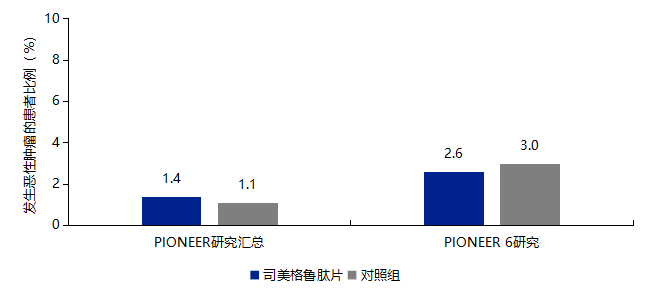
-
Semaglutide tablets are contraindicated in patients with a history or family history of medullary thyroid carcinoma and in patients with type 2 multiple endocrine neoplasia syndrome. -
Patients should be informed of the potential risk of medullary thyroid carcinoma and the symptoms of thyroid tumors.
3.3 No Increased Risk of Pancreatitis with Semaglutide Tablets
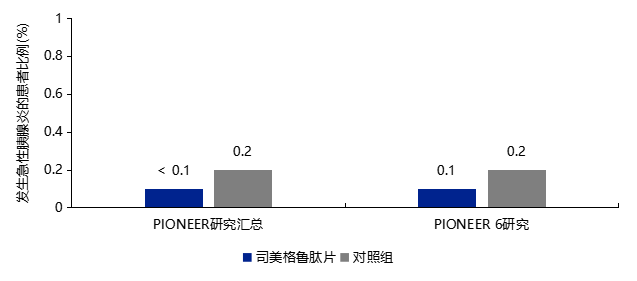
-
Patients with a history of pancreatitis should use semaglutide tablets with caution. -
If pancreatitis is suspected, semaglutide tablets should be discontinued; once pancreatitis is confirmed, semaglutide tablets should not be used for treatment. -
In clinical practice, patients should be informed of the characteristic symptoms of acute pancreatitis.
3.4 No Increased Risk of Gallbladder Disease with Semaglutide Tablets
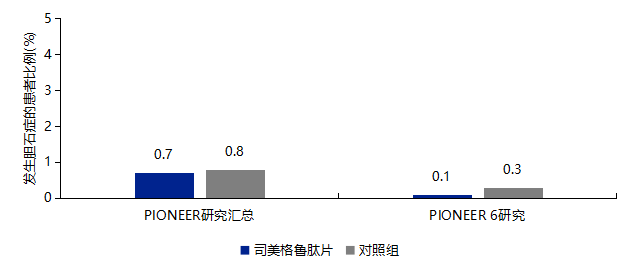
-
If gallstones are suspected, gallbladder examination and appropriate clinical follow-up are required. -
Prior to medication, enhance communication and guidance with patients, informing them of the potential risks of gallstones or cholecystitis, and guiding them to seek timely medical attention in case of suspected gallstones or cholecystitis.
3.5 Semaglutide Tablets Have Confirmed Cardiovascular Safety
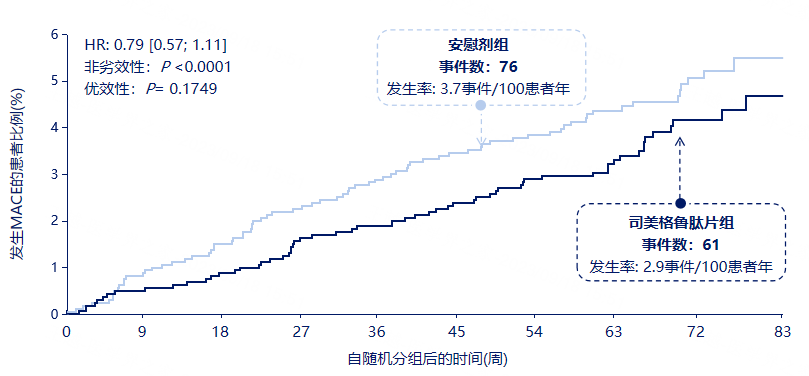
3.6 No Increased Risk of Diabetic Retinopathy with Semaglutide Tablets (DR) Safety Signals
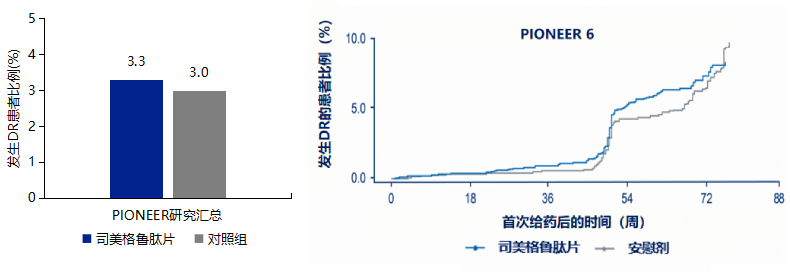
-
Patients with a history of DR should monitor the progression of DR during treatment with semaglutide tablets. -
If patients experience changes in vision during treatment with semaglutide tablets, they should seek medical attention promptly.
3.7 No Increased Risk of Renal Adverse Events with Semaglutide Tablets
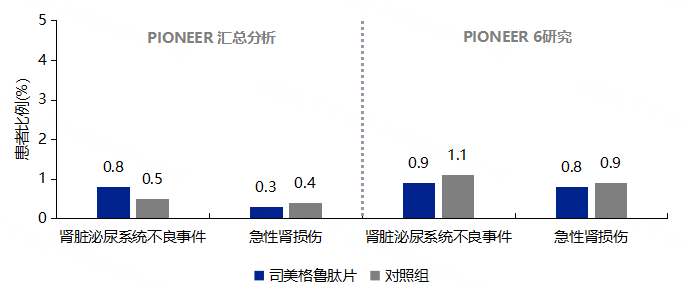
Note: The summary of PIONEER studies includes data from PIONEER 1-5, 7-10, with the control group including active control drugs and placebo; PIONEER 6 control group is placebo.
-
No dosage adjustment is needed for patients with mild to moderate renal impairment; currently, experience in using semaglutide tablets in patients with severe renal impairment is limited. -
Semaglutide tablets are not recommended for use in patients with end-stage renal disease. -
Patients experiencing severe gastrointestinal adverse reactions during treatment should have their renal function monitored.
Conclusion

Chen Lulu
-
Department of Endocrinology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology
-
Associate Professor, Chief Physician, Doctor of Medicine, Doctoral Supervisor, Expert enjoying special government allowances from the State Council.
-
Vice Chairman of the 10th Committee of the Endocrinology Society of the Chinese Medical Association;
-
Vice Chairman of the Diabetes Professional Committee of the Chinese Women Physicians Association;
-
First batch of specially invited experts from the Center for Drug Evaluation of the National Medical Products Administration;
-
Former Chairman of the Hubei Diabetes Society;
-
Chairman of the 4th and 5th Hubei Endocrinology Society;
-
Vice Chairman of the Cardiovascular Metabolic Disease Physician Committee of the Hubei Preventive Medicine Association;
-
Director of the Hubei Clinical Medical Research Center for Diabetes and Metabolic Diseases;
-
Director of the Quality Control Center for Endocrinology and Diabetes in Hubei Province.
-
Involved in key projects of the National Key Research and Development Program, multiple National Natural Science Foundation projects, the 11th Five-Year Plan Support Project, and major projects of the Hubei Provincial Science and Technology Department. Published 411 articles and professional papers in domestic and international academic journals, including 108 SCI articles. Awarded first and second prizes for scientific and technological progress in Hubei Province and the first prize for the Chinese Medical Science and Technology Award. Recognized as one of the top ten medical influencers in China in 2021.


