1. Analysis of physicochemical properties of pure and adulterated honey.
2. Determination of sugar content in pure and adulterated honey using high-performance liquid chromatography.
3. Identification of pure and adulterated honey based on FTIR-ATR and PLS regression.
The combination of Fourier transform infrared spectroscopy (FTIR) and attenuated total reflectance (ATR) techniques with chemometric models was applied for the rapid identification and quantitative analysis of adulterants in honey. The study subjects included bee honey and stingless bee honey, with acidic adulterants (such as acetic acid, citric acid, and tamarind extract) and sugar adulterants (such as liquid corn syrup, sucrose, palm sugar, and glucose) added in proportions of 1%, 3%, 5%, and 7%, with concentrations of 1%, 2%, and 3%, respectively. Before conducting FTIR tests, physicochemical properties (including Brix, moisture content, pH, and free acidity) and sugar composition (glucose, fructose, and sucrose) were determined for each honey sample and their adulterants. FTIR spectral data were obtained in the mid-infrared region (400-3600 cm^-1) for all samples, and partial least squares (PLS) was used to process the obtained spectra, aiming to construct a predictive model capable of effectively identifying honey adulterants. The results indicated that the established PLS model had a high coefficient of determination for samples containing acetic acid (R²=0.95), citric acid (R²=0.98), tamarind extract (R²=0.97), and liquid corn syrup in bee honey (R²=0.99). Additionally, the effectiveness of the mixture detection was further confirmed through physicochemical methods. In summary, the combined application of FTIR-ATR spectroscopy and PLS regression analysis can serve as an efficient, accurate, and non-destructive method for assessing honey authenticity.
Honey is synthesized by bees through the collection and transformation of plant nectar, secretions from living plant parts, or excretions from plant-feeding insects. Bees store these substances in hives, allowing them to mature gradually. The main nutritional components of honey include water, carbohydrates (with fructose accounting for about 38% and glucose for about 31%), as well as proteins, enzymes, free amino acids, minerals, vitamins, organic acids, and phenolic compounds as minor components. The nutritional value, particle size, taste, and texture of honey are influenced by its moisture content, reducing sugars, free acids, conductivity, sucrose concentration, and hydroxymethylfurfural (HMF) content. Phytochemicals present in honey, such as phenolic compounds, coumarin, spathulenol, lunamarin, flavonoids, and proanthocyanidins, are crucial for determining its antioxidant activity. This activity endows honey with anti-inflammatory, anticancer, antithrombotic, and anti-atherosclerotic properties (Ciursă et al., 2021). Due to the unique flavor and nutritional advantages conferred by these chemical components, honey typically commands a higher market price than other sweeteners like sucrose syrup. However, there are significant variations in honey quality and cost. Unfortunately, as a high-quality nutritional and medicinal food, honey is sometimes directly affected by adulteration, leading to the degradation of its unique bioactive components. Adulteration refers to the deliberate addition of low-value substances to pure products to degrade, diminish quality, or replace high-value substances.
Honey adulteration is a form of fraud, where adulterers typically add low-quality and inexpensive sugars or acidic substances to honey for illicit profit. However, the methods of adulteration in the honey market extend far beyond what this study covers; we selected the most common and frequently reported adulterants from the literature. Common adulterants include but are not limited to commercial syrups such as cane sugar syrup, palm sugar, corn syrup, and sucrose syrup, as well as low-cost food additives such as citric acid, vinegar, and tamarind extract, which are classified as GRAS (generally recognized as safe) (Mwale, 2023). Additionally, substances such as ripe bananas, molasses, corn and/or wheat flour syrup, and sweet potato powder/syrup (Damto, Birhanu, & Zewdu, 2023; Damto, Zewdu, & Birhanu, 2024; Gemeda & Negera, 2017) have also been used for honey adulteration, although their market share is relatively small and primarily concentrated in specific regions. Adulteration not only alters the chemical composition of honey but also reduces the concentration of bioactive substances with health benefits, thereby diminishing the overall quality and market value of honey. For example, there is a high-end product known as stingless bee honey, famous for its unique sour taste, while regular bee honey is a more economical choice, tasting sweeter. Repaeh, Zhi, and Long (2022) noted that stingless bee honey (SBH) is favored for its unique sour taste, aroma, and higher price, in stark contrast to the nectar produced by ordinary bees (A. mellifera). The common adulterants found in SBH include glucose syrup, fructose syrup, and nectar. According to local beekeepers, SBH adulteration often involves diluting honey with water to increase yield, thus illegally profiting. In some cases, cheaper honey like FH is mixed with an acidic liquid (such as vinegar) to mimic the sour characteristics of SBH and is then sold at prices close to pure SBH. Therefore, some unscrupulous merchants adjust the flavor of stingless bee honey by adding slightly acidic substances and sell it at a higher price than ordinary bee honey (Rafi Fakhlaei et al., 2024). On the other hand, some producers mix cheap sugars into Itama honey to increase yield, economic benefits, and meet consumer demand (Rafieh Fakhlaei et al., 2021). Unfortunately, such adulteration practices may have negative impacts on human health and require in-depth study. In light of this, international agencies and organizations are actively taking measures to strengthen honey quality monitoring systems and establish relevant safety assurance measures. There has been extensive discussion in the existing literature regarding methods for assessing honey quality and authenticity (Wu et al., 2023; Zhang and Abdulla, 2022b).
Various traditional methods can be used for honey authenticity testing, such as thin-layer chromatography (TLC), which has been mainly used since the late 1970s to detect the presence of high fructose corn syrup in honey. However, the application of this method is somewhat limited due to possible false-positive results from sugar hydrolysis (Wu et al., 2017). Additionally, polymerase chain reaction (PCR) has also been considered a traditional means to verify honey authenticity (Sobrino-Gregorio et al., 2019). However, these methods struggle to accurately distinguish adulterated products that closely resemble pure honey in color, viscosity, aroma, and taste (Price et al., 2019). Therefore, traditional testing methods are generally only suitable for preliminary screening and should not be used as the final determination. With advances in technology, traditional adulteration testing techniques are gradually being phased out, serving only as auxiliary references. Meanwhile, modern spectroscopic techniques, including high-performance liquid chromatography (HPLC) (Zhang G. and Abdulla, 2022a), nuclear magnetic resonance (NMR) (Kalyani, Mestri, and Consogni, 2022), and stable carbon isotope ratio mass spectrometry (SCIRA) (Pérez, Raven, Harold, and Echevestre, 2024), have been widely used in the field of honey authenticity testing. Nevertheless, HPLC may encounter measurement errors due to high sucrose and maltose content when dealing with adulterated syrups (Akas, 2023). NMR systems have lower sensitivity and are complex to operate, requiring a high level of expertise and technical knowledge (Izenaka, Enso-Atingdana, and Zhang, 2019). Notably, while SCIRA analysis can effectively detect honey mixed with C4 plant sources (such as sucrose syrup, fructose syrup, corn syrup, and maltose syrup), it is powerless against adulterants derived from C3 plants (such as rice syrup and beet syrup) (Xie Hata et al., 2024).
Therefore, developing rapid, non-destructive, user-friendly, and sensitive analytical techniques is particularly important. Vibrational spectroscopy can provide reliable structural and fingerprint information for unique compounds present in various substances, making it an effective tool for verifying food authenticity. As a non-destructive fingerprinting method, vibrational spectroscopy enables rapid analysis. For instance, Fourier transform infrared spectroscopy (Brar et al., 2023), Fourier transform Raman spectroscopy (X. Wu et al., 2023), and near-infrared spectroscopy (G. Zhang & Abdulla, 2022b) have shown great potential in the field of food identification, especially in honey adulteration detection. Compared to other analytical techniques, these methods are widely regarded as having significant advantages due to their efficiency in quantitative and qualitative analysis, minimal sample requirements, environmental friendliness, and little or no sample pretreatment (Rafieh Fakhlaei, Babadi et al., 2024). Raman spectroscopy has proven to be an effective tool for detecting honey adulteration, particularly when used in conjunction with other analytical techniques. However, Raman spectroscopy may lack sensitivity for low-dose or complex components of adulterants. For example, the strong Raman signal of water molecules may interfere with the detection of adulterants, especially in honey samples with high moisture content. Fluorescence spectroscopy, due to its specificity and simplicity, is suitable for identifying specific adulterants in honey. However, this method also has limitations, as pollen and pigments in honey may interfere with the fluorescence signal, increasing detection difficulty. Especially when dealing with viscous samples like honey, the sample preparation process can be time-consuming. Fourier transform infrared spectroscopy (FTIR) offers a rapid, non-destructive solution for chemical measurement, suitable for qualitative analysis. By utilizing narrow spectral bands in the fingerprint region to reduce overlap issues, simple mathematical processing methods can be employed, such as establishing calibration relationships between peak height or area and concentration (Rios-Corripio, Rojas-Lopez∗, & Delgado-Macuil, 2012). However, in complex systems like adulterated honey samples, the highly similar spectra of various components necessitate more complex processing techniques. Multivariate statistical methods, such as partial least squares (PLS), provide strong support for constructing predictive models related to the content of multiple adulterants in honey (Prata & da Costa, 2024).
Fourier transform infrared spectroscopy (FTIR) is an optical sensing technology based on the characteristic absorption of specific chemical groups in the infrared (IR) region of food components (Brar et al., 2023). When combined with chemometric analysis, this method not only expands sample information but also significantly enhances model accuracy. In recent years, this approach has been widely applied in the field of food safety analysis. For example, Riswahyuli, Rohman, Setyabudi, and Raharjo (2020) successfully detected adulteration in Indonesian wild honey using FTIR-ATR combined with a partial least squares (PLS) model. Experimental results showed that commercial sugars, including sucrose, coconut sugar, and cane sugar, were present in wild honey, with corresponding predicted R² values of 0.9977, 0.9983, and 0.9946, respectively. Furthermore, according to research by Damto et al. (2023), the use of FTIR analysis combined with multivariate statistical analysis achieved a cumulative variance of 96% for two principal components, demonstrating that the parameters studied could effectively differentiate pure honey from adulterated honey. Notably, the FTIR technique greatly accelerates the sample analysis process due to its lack of sample pretreatment and suitability for small sample sizes. By combining Fourier transform infrared spectroscopy (FTIR) with multivariate statistical techniques, it is possible to directly, reliably, and efficiently obtain specific information on multiple parameters (Damto, Zewdu, & Birhanu, 2023). Data analysis of FTIR spectral technology relies on advanced statistical programs, with partial least squares (PLS) serving as a chemometric tool for accurately identifying subtle differences between pure and adulterated honey samples.
Using a single spectral technique to detect honey adulteration may result in lower accuracy and robustness of the established detection model than expected, primarily due to a lack of necessary relevant information in the data source. Therefore, this experiment attempts to combine Fourier transform infrared spectroscopy (FTIR) data with partial least squares regression (PLS-R) to construct a more precise and stable honey adulteration detection model. Previous studies have mainly focused on single types of honey and their adulterants (such as sugars or acids). However, in this study, we utilized FTIR spectroscopy combined with PLS-R to systematically explore the effects of common acidic and sugary substances on two different sources of honey—bee honey and non-bee honey (SBH). The goal of this study is to construct a comprehensive database of honey adulteration, providing valuable data support for future honey authenticity testing.
4. Results and Discussion
1. Physicochemical Parameter Analysis
The physicochemical parameters (including sugar degree, moisture content, pH, and free acidity) of pure honey, adulterated honey, and SBH were measured, and the sugar composition was determined using high-performance liquid chromatography (HPLC). The physicochemical analysis results of pure honey and adulterated honey are summarized in Table 1, while the physicochemical analysis results of pure honey and adulterated SBH are summarized in Table 2. The soluble solid content (SSC) of pure honey was 78.15±0.78°Brix, while the SSC of pure SBH was 65.40±0.10°Brix. The SSC value of pure honey reported by Rivera-Mondragon et al. (2023) was 79.30±1.30°Brix, which is close to the results of this study. However, the SSC value of SBH is reported to range from 64.5 to 75.8°Brix, which may relate to its higher moisture content and lower sugar content (Raypah et al., 2022). The SSC of acid-adulterated honey significantly decreased (p<0.05) with increasing acid concentration (see Table 1). At all concentrations, the acetic acid-adulterated honey exhibited significantly (p<0.05) lower sugar degree compared to pure honey.
Generally, honey exhibits a naturally acidic characteristic, primarily due to its moisture content. Honey with higher moisture content is more prone to fermentation, leading to increased free acidity and decreased pH. For example, SBH samples had higher moisture content than ordinary honey samples. Therefore, the pH of SBH samples was lower than that of ordinary honey. The results of this study showed that the free acidity of SBH samples was 100.33 meq/kg, approximately 1.7 times higher than that of ordinary honey samples (60.00 meq/kg) (see Table 1, Table 2). This result aligns with the findings of Esa et al. (2022), who noted that SBH has higher free acidity than ordinary honey. Thus, it can be confirmed that SBH is more acidic than ordinary honey. Literature indicates that the primary determinant of honey acidity is the content of its internal organic acids (Valverde et al., 2022). However, for adulterated honey samples, the free acidity significantly increased (p < 0.05) and intensified with rising acid concentrations.
2. Sugar Content in Honey
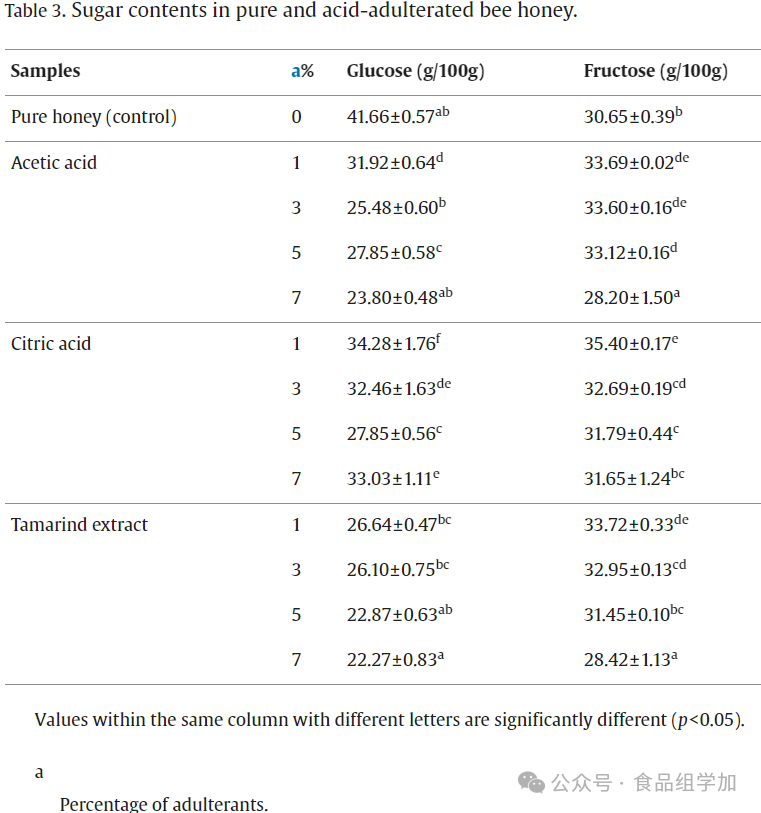 In this study, the sugar composition of pure honey, adulterated honey, and SBH was quantitatively analyzed using high-performance liquid chromatography (HPLC). Table 3 lists the average values ± standard deviation of glucose and fructose content in pure and adulterated honey. The results showed that the average glucose and fructose content in pure honey was 41.66±0.57 g/100 g and 30.65±0.39 g/100 g, respectively. In pure SBH, the average glucose and fructose content was 23.15±0.14 g/100 g and 25.06±0.53 g/100 g (see Table 4). In comparison, the glucose and fructose content in pure honey was significantly higher than in pure SBH. For acid-adulterated honey, the average glucose and fructose content significantly decreased (p<0.05) with increasing acid concentration. Conversely, in sugar-adulterated SBH, the glucose and fructose content significantly increased (p<0.05) with increasing concentration. These findings further confirm the possible presence of other types of sugar adulterants in SBH. Furthermore, the total glucose and fructose content in honey reached 72.31 g/100 g, meeting the standards set by the Codex Alimentarius Commission (2001), which stipulates not less than 60 g/100 g. Notably, the total glucose and fructose content in SBH was 48.21 g/100 g. Nevertheless, experimental data still indicate that the main monosaccharide components in both pure honey and SBH are fructose and glucose, a conclusion consistent with the findings of F. R. et al. (2020), thus verifying the authentic nature of honey.
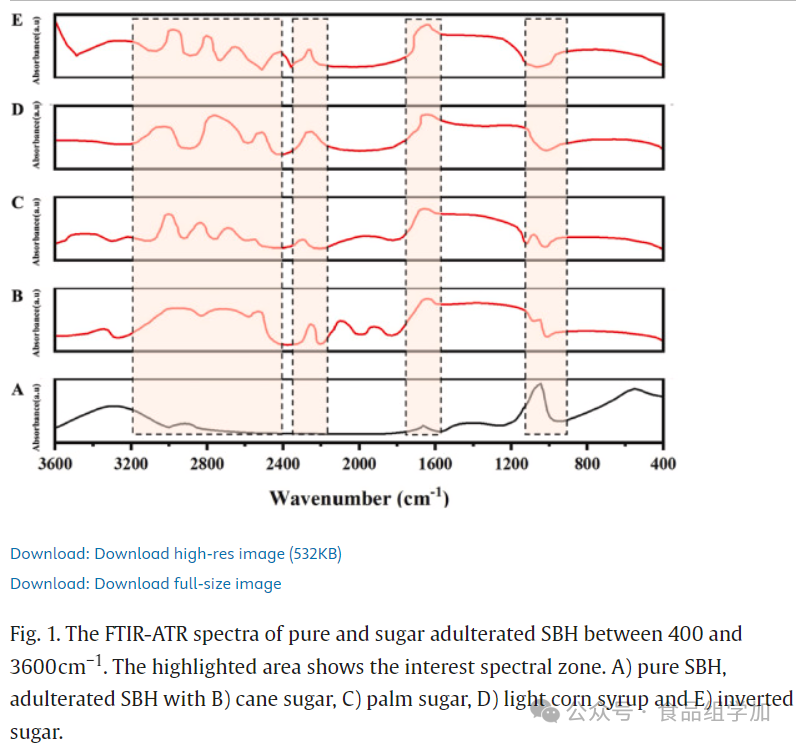
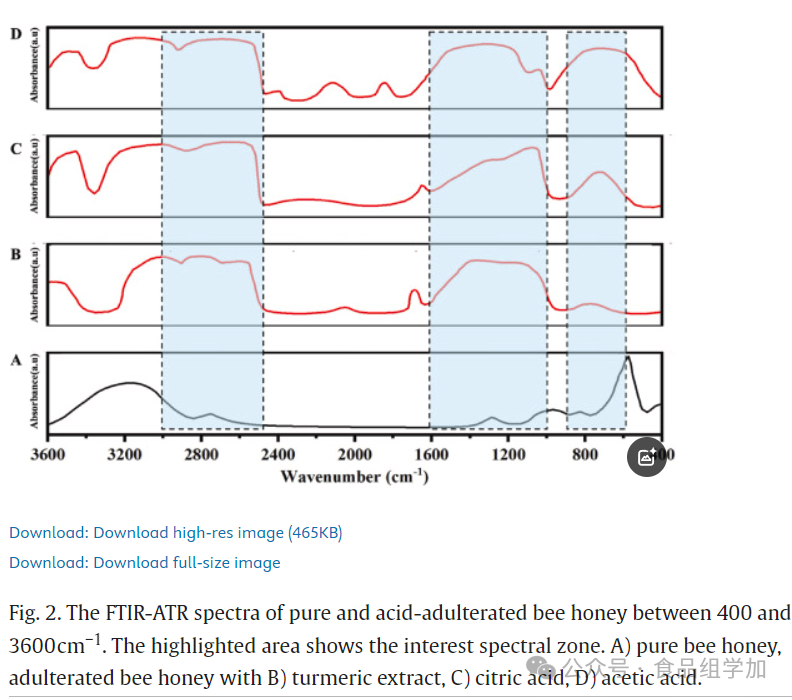
A detailed analysis of the infrared spectra (400-3600 cm^-1) of pure honey, adulterated honey (containing acids and sugars), and their mixtures (SBH) was conducted (see Figures 1, 2). Table S1 lists the functional group vibration bandwidths corresponding to identified compounds. Figure 1 shows the spectral data of pure honey and sugar-adulterated SBH. In the range of 900-1800 cm^-1, the carbon-oxygen and carbon-carbon stretching modes (900-1000 cm^-1) and the carbon=oxygen stretching (1600-1700 cm^-1) are the main distinguishing factors, as these regions directly reflect the sugar content in the samples. The range of 2400-3200 cm^-1 is associated with the O-H stretching of carboxylic acids in honey. SBH and all sugar-adulterated samples exhibited similar spectral characteristics in the 400-1800 cm^-1 range. The spectral bands in the 2800-3000 cm^-1 range represent the C-H stretching vibrations in the sugar molecule backbone, including aromatic and alkyl chains. These vibrations correspond to the alternating symmetric and asymmetric stretching of functional groups -CH2 and -CH3 (Aliano-Gonzalez et al., 2019). Additionally, signals of C-C-H and H-C-O deformation were detected near 1700 cm^-1. Given the high sugar content in honey (Cardenas-Escudero et al., 2023), the presence of such vibrations is reasonable. The formation of carboxylic acid dimers promotes the establishment of strong hydrogen bonds, leading to a series of vibrations originating from -OH groups. A significant peak at 1640 cm^-1 indicates the deformation vibration of -OH groups (Damto et al., 2023). Peaks in the 1600-1700 cm^-1 range are attributed to the C=O stretching vibrations of sugars and the N-H bending vibrations of amide I components in proteins. In the range of 930-1040 cm^-1, significant spectral enhancement was also observed due to the enhancement of C-O and C-C stretching vibrations in the carboxyl -OH groups and sugar structures. Figure 2 reveals that the spectral graphs of pure honey and adulterated honey are highly consistent in the 600-900 cm^-1, 1000-1600 cm^-1, and 2500-3000 cm^-1 ranges. The 700-950 cm^-1 range is considered the cyclic region of carbohydrates, consisting of two modes of asymmetric stretching vibrations of C-O-C, corresponding to the α and β isomers, providing additional information about the sample components (Kozłowicz et al., 2020). Entering the fingerprint region (1000-1600 cm^-1), this area is characterized by various vibration modes of carbohydrates and ketones, with signals at 1020 and 1343 cm^-1 attributable to the C-OH, C-H, and C-O stretching vibrations of carbohydrates (i.e., sugars) (Ciursă et al., 2021). Peaks in the 2600-3000 cm^-1 range correspond to the vibration modes of carbohydrates (sugars), amino acids (Cardenas-Escudero et al., 2023), and carboxylic acids (Ciursă et al., 2021). A significant peak appears in the 3000-3650 cm^-1 range, especially prominent around 3335 cm^-1, which is related to the stretching vibrations of -OH groups in carbohydrates, water, and organic acids (Kozłowicz et al., 2020). This region is often associated with the stretching vibrations of carboxylic acids and the -NH3 stretching bands of free amino acids, showing slight enhancement. The vibrations observed near 3335 cm^-1 may originate from carboxylic acids. Due to the broadband characteristics of -OH group vibrations, their absorption significantly enhances the stretching vibrations of C-H bonds in -CH2 and -CH3 groups. The wavenumber variations observed in the 3335 cm^-1 and 930 cm^-1 ranges are particularly significant in pure honey, adulterated honey, and SBH.
In this study, the sugar composition of pure honey, adulterated honey, and SBH was quantitatively analyzed using high-performance liquid chromatography (HPLC). Table 3 lists the average values ± standard deviation of glucose and fructose content in pure and adulterated honey. The results showed that the average glucose and fructose content in pure honey was 41.66±0.57 g/100 g and 30.65±0.39 g/100 g, respectively. In pure SBH, the average glucose and fructose content was 23.15±0.14 g/100 g and 25.06±0.53 g/100 g (see Table 4). In comparison, the glucose and fructose content in pure honey was significantly higher than in pure SBH. For acid-adulterated honey, the average glucose and fructose content significantly decreased (p<0.05) with increasing acid concentration. Conversely, in sugar-adulterated SBH, the glucose and fructose content significantly increased (p<0.05) with increasing concentration. These findings further confirm the possible presence of other types of sugar adulterants in SBH. Furthermore, the total glucose and fructose content in honey reached 72.31 g/100 g, meeting the standards set by the Codex Alimentarius Commission (2001), which stipulates not less than 60 g/100 g. Notably, the total glucose and fructose content in SBH was 48.21 g/100 g. Nevertheless, experimental data still indicate that the main monosaccharide components in both pure honey and SBH are fructose and glucose, a conclusion consistent with the findings of F. R. et al. (2020), thus verifying the authentic nature of honey.
A detailed analysis of the infrared spectra (400-3600 cm^-1) of pure honey, adulterated honey (containing acids and sugars), and their mixtures (SBH) was conducted (see Figures 1, 2). Table S1 lists the functional group vibration bandwidths corresponding to identified compounds. Figure 1 shows the spectral data of pure honey and sugar-adulterated SBH. In the range of 900-1800 cm^-1, the carbon-oxygen and carbon-carbon stretching modes (900-1000 cm^-1) and the carbon=oxygen stretching (1600-1700 cm^-1) are the main distinguishing factors, as these regions directly reflect the sugar content in the samples. The range of 2400-3200 cm^-1 is associated with the O-H stretching of carboxylic acids in honey. SBH and all sugar-adulterated samples exhibited similar spectral characteristics in the 400-1800 cm^-1 range. The spectral bands in the 2800-3000 cm^-1 range represent the C-H stretching vibrations in the sugar molecule backbone, including aromatic and alkyl chains. These vibrations correspond to the alternating symmetric and asymmetric stretching of functional groups -CH2 and -CH3 (Aliano-Gonzalez et al., 2019). Additionally, signals of C-C-H and H-C-O deformation were detected near 1700 cm^-1. Given the high sugar content in honey (Cardenas-Escudero et al., 2023), the presence of such vibrations is reasonable. The formation of carboxylic acid dimers promotes the establishment of strong hydrogen bonds, leading to a series of vibrations originating from -OH groups. A significant peak at 1640 cm^-1 indicates the deformation vibration of -OH groups (Damto et al., 2023). Peaks in the 1600-1700 cm^-1 range are attributed to the C=O stretching vibrations of sugars and the N-H bending vibrations of amide I components in proteins. In the range of 930-1040 cm^-1, significant spectral enhancement was also observed due to the enhancement of C-O and C-C stretching vibrations in the carboxyl -OH groups and sugar structures. Figure 2 reveals that the spectral graphs of pure honey and adulterated honey are highly consistent in the 600-900 cm^-1, 1000-1600 cm^-1, and 2500-3000 cm^-1 ranges. The 700-950 cm^-1 range is considered the cyclic region of carbohydrates, consisting of two modes of asymmetric stretching vibrations of C-O-C, corresponding to the α and β isomers, providing additional information about the sample components (Kozłowicz et al., 2020). Entering the fingerprint region (1000-1600 cm^-1), this area is characterized by various vibration modes of carbohydrates and ketones, with signals at 1020 and 1343 cm^-1 attributable to the C-OH, C-H, and C-O stretching vibrations of carbohydrates (i.e., sugars) (Ciursă et al., 2021). Peaks in the 2600-3000 cm^-1 range correspond to the vibration modes of carbohydrates (sugars), amino acids (Cardenas-Escudero et al., 2023), and carboxylic acids (Ciursă et al., 2021). A significant peak appears in the 3000-3650 cm^-1 range, especially prominent around 3335 cm^-1, which is related to the stretching vibrations of -OH groups in carbohydrates, water, and organic acids (Kozłowicz et al., 2020). This region is often associated with the stretching vibrations of carboxylic acids and the -NH3 stretching bands of free amino acids, showing slight enhancement. The vibrations observed near 3335 cm^-1 may originate from carboxylic acids. Due to the broadband characteristics of -OH group vibrations, their absorption significantly enhances the stretching vibrations of C-H bonds in -CH2 and -CH3 groups. The wavenumber variations observed in the 3335 cm^-1 and 930 cm^-1 ranges are particularly significant in pure honey, adulterated honey, and SBH.
 4. PLS Model Predictions for Adulterated Honey
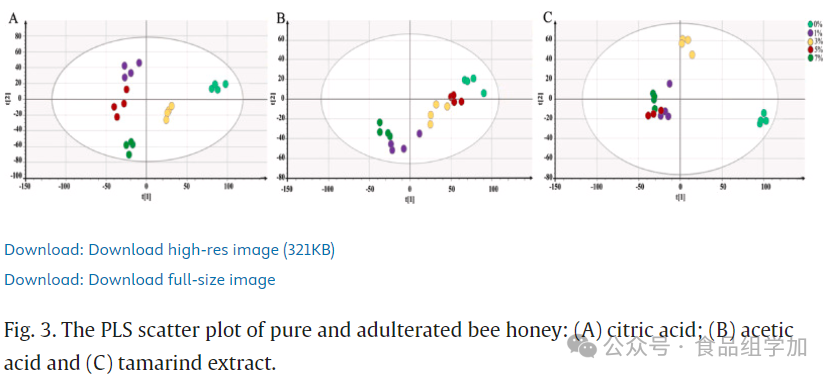
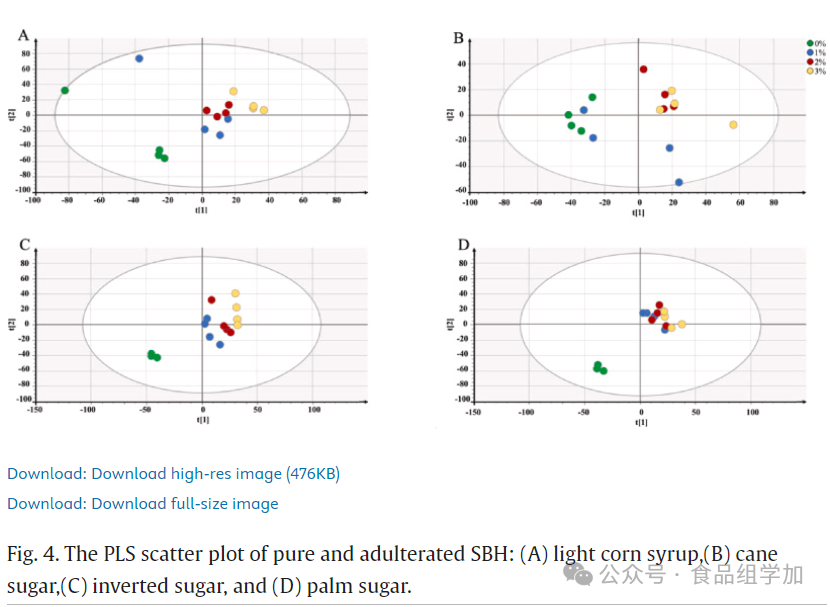
The FTIR spectral data were used to establish models for various adulterants through partial least squares (PLS) multivariate analysis. These models revealed the relationships between independent variables (FTIR-ATR spectral data) and dependent variables (adulterant concentrations). Through score scatter plots, pure honey and adulterated honey could be effectively distinguished, as shown in Figures 3 and 4. All pure honey and adulterated honey samples were sorted based on PLS components 1 and 2. The adulterated honey samples significantly deviated from the pure honey samples and did not fall within the same component range as pure honey. These findings align with those of Dranca et al. (2022), who pointed out that when the adulteration concentration of honey reached 5%, its characteristics were similar to pure honey, while at 10% and 20% adulteration concentrations, the honey samples were clearly distinguishable from others.
4. PLS Model Predictions for Adulterated Honey
The FTIR spectral data were used to establish models for various adulterants through partial least squares (PLS) multivariate analysis. These models revealed the relationships between independent variables (FTIR-ATR spectral data) and dependent variables (adulterant concentrations). Through score scatter plots, pure honey and adulterated honey could be effectively distinguished, as shown in Figures 3 and 4. All pure honey and adulterated honey samples were sorted based on PLS components 1 and 2. The adulterated honey samples significantly deviated from the pure honey samples and did not fall within the same component range as pure honey. These findings align with those of Dranca et al. (2022), who pointed out that when the adulteration concentration of honey reached 5%, its characteristics were similar to pure honey, while at 10% and 20% adulteration concentrations, the honey samples were clearly distinguishable from others.
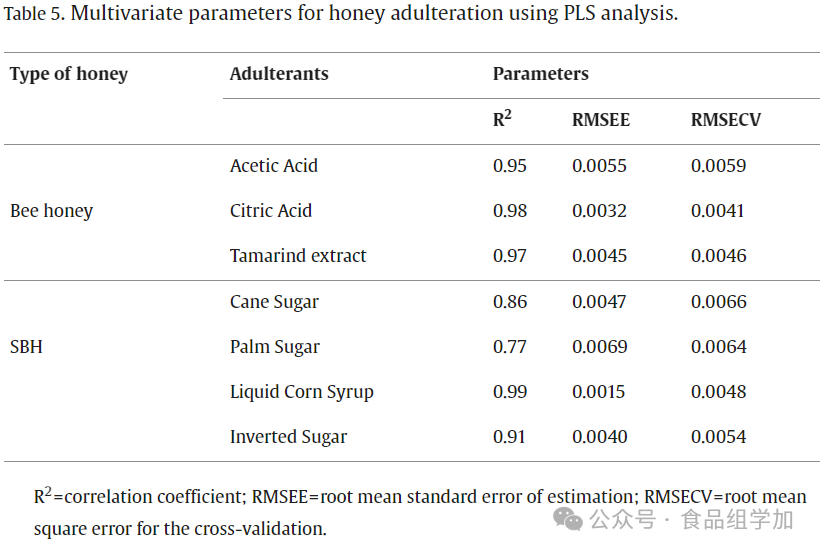
 The R² value reflects the degree of correlation between actual observed values and model predicted values; when R² equals 1, it indicates a complete correlation between the validation set data. Based on the research by Anguebes-Franseschi et al. (2019), an ideal model should have the lowest calibration standard error, with the maximum allowable error not exceeding 3. Table 5 lists the partial least squares (PLS) models established for adulterated honey and SBH. The results showed that for honey samples adulterated with acetic acid, citric acid, and orange juice, the PLS models achieved high R² values (all above 0.9), specifically 0.95, 0.98, and 0.97, respectively. Similarly, when dealing with SBH containing different types of sugars, the PLS models also exhibited high R² values (all exceeding 0.9), with liquid corn syrup and sucrose corresponding to R² values of 0.99 and 0.91, while palm sugar was 0.77. The RMSEE and RMSECV indicators for all PLS models ranged from 0.0032 to 0.0055 (for acetic acid-adulterated honey) and 0.0015 to 0.0069 (for sugar-adulterated SBH), all of which are below the set maximum threshold of 3. According to the research by Elhamdaoui et al. (2020), the better the predictive performance of the PLS model, the lower the values of RMSEE and RMSECV. In summary, the above evaluation indicators confirm the excellent performance of the model in predicting adulterants in honey and SBH.
The R² value reflects the degree of correlation between actual observed values and model predicted values; when R² equals 1, it indicates a complete correlation between the validation set data. Based on the research by Anguebes-Franseschi et al. (2019), an ideal model should have the lowest calibration standard error, with the maximum allowable error not exceeding 3. Table 5 lists the partial least squares (PLS) models established for adulterated honey and SBH. The results showed that for honey samples adulterated with acetic acid, citric acid, and orange juice, the PLS models achieved high R² values (all above 0.9), specifically 0.95, 0.98, and 0.97, respectively. Similarly, when dealing with SBH containing different types of sugars, the PLS models also exhibited high R² values (all exceeding 0.9), with liquid corn syrup and sucrose corresponding to R² values of 0.99 and 0.91, while palm sugar was 0.77. The RMSEE and RMSECV indicators for all PLS models ranged from 0.0032 to 0.0055 (for acetic acid-adulterated honey) and 0.0015 to 0.0069 (for sugar-adulterated SBH), all of which are below the set maximum threshold of 3. According to the research by Elhamdaoui et al. (2020), the better the predictive performance of the PLS model, the lower the values of RMSEE and RMSECV. In summary, the above evaluation indicators confirm the excellent performance of the model in predicting adulterants in honey and SBH.
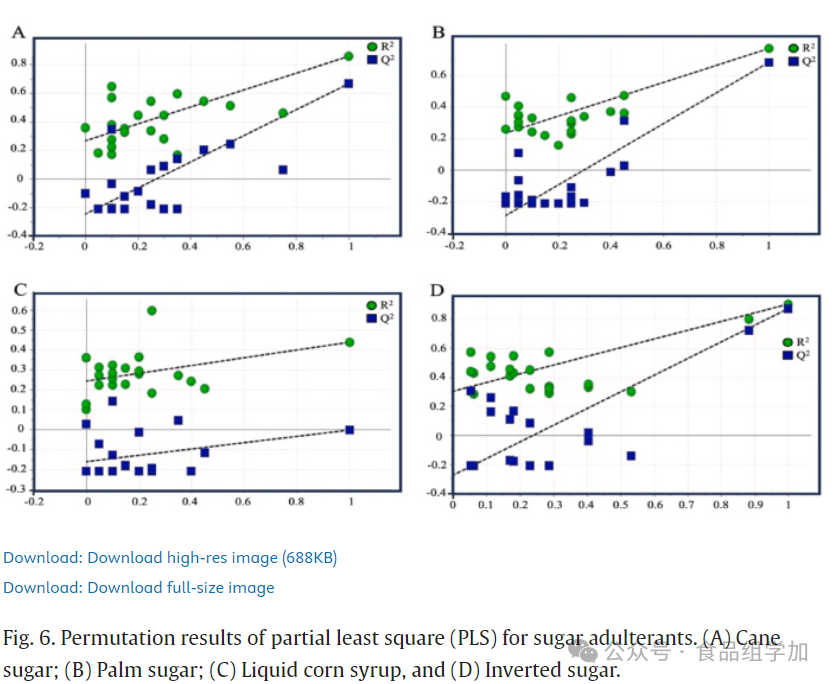
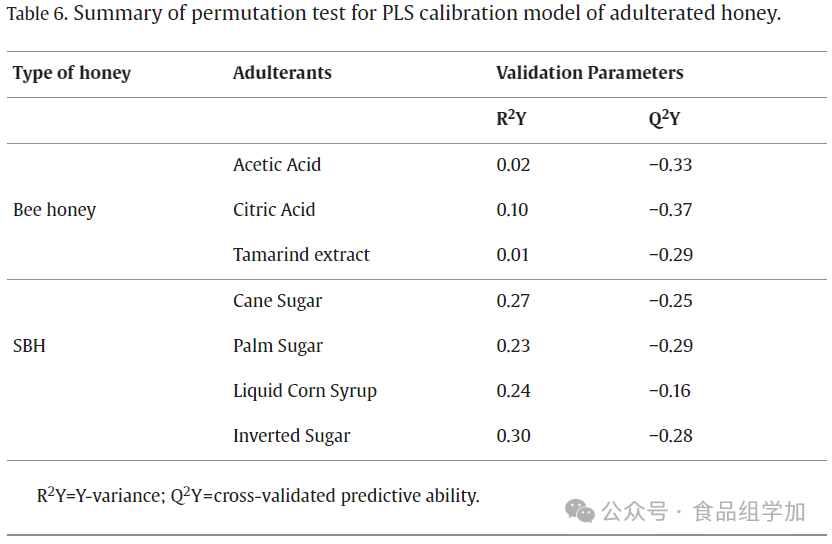
 5. Validation of the PLS Model
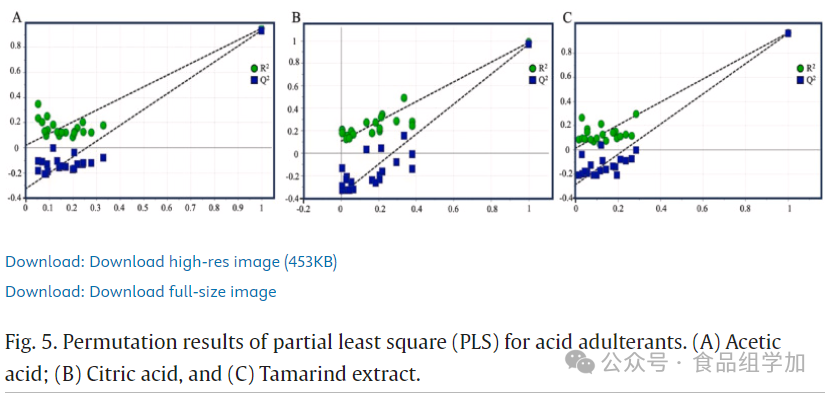
The PLS model was evaluated for its predictive performance through cross-validation and effectively prevented data overfitting. A permutation test was used to validate the accuracy of multivariate calibration. Figures 5 and 6 display the original model’s permutation parameters and their R² and Q² values. The X-axis represents the correlation coefficients between the original model and the permutation model, while the Y-axis shows the R² (goodness of fit) and Q² (predictive ability) values. During the validation process, the performance of the original model significantly exceeded that of the permutation model. The X-axis reflects the correlation between the original data and the permutation data responses, while the Y-axis displays the R² and Q² values. In this study, the PLS model underwent cross-validation through 20 permutation tests. According to the research by Formosa et al. (2020), ideally, the intercept values of R²Y and Q²Y in the permutation test should not exceed 0.4 and 0.05. Based on the data in Table 6, the intercept values of R²Y and Q²Y for acid-adulterated honey and sugar-adulterated SBH were both below 0.4 and 0.05. This indicates that all constructed PLS models did not experience overfitting. Furthermore, these results align with our previous findings regarding the toxicological assessment of adulterated SBH (Rafieh Fakhlaei et al., 2021). This study proposes an advanced analytical method that combines FTIR-ATR with PLSR for honey authenticity and quality analysis. Given that PLSR can handle collinearity issues, reduce dimensionality, and avoid overfitting, it becomes an effective tool for addressing limited sample sizes. In this study, the PLSR algorithm was specifically employed to handle outliers and noise in the model.
5. Validation of the PLS Model
The PLS model was evaluated for its predictive performance through cross-validation and effectively prevented data overfitting. A permutation test was used to validate the accuracy of multivariate calibration. Figures 5 and 6 display the original model’s permutation parameters and their R² and Q² values. The X-axis represents the correlation coefficients between the original model and the permutation model, while the Y-axis shows the R² (goodness of fit) and Q² (predictive ability) values. During the validation process, the performance of the original model significantly exceeded that of the permutation model. The X-axis reflects the correlation between the original data and the permutation data responses, while the Y-axis displays the R² and Q² values. In this study, the PLS model underwent cross-validation through 20 permutation tests. According to the research by Formosa et al. (2020), ideally, the intercept values of R²Y and Q²Y in the permutation test should not exceed 0.4 and 0.05. Based on the data in Table 6, the intercept values of R²Y and Q²Y for acid-adulterated honey and sugar-adulterated SBH were both below 0.4 and 0.05. This indicates that all constructed PLS models did not experience overfitting. Furthermore, these results align with our previous findings regarding the toxicological assessment of adulterated SBH (Rafieh Fakhlaei et al., 2021). This study proposes an advanced analytical method that combines FTIR-ATR with PLSR for honey authenticity and quality analysis. Given that PLSR can handle collinearity issues, reduce dimensionality, and avoid overfitting, it becomes an effective tool for addressing limited sample sizes. In this study, the PLSR algorithm was specifically employed to handle outliers and noise in the model.
 This study proposes a method combining FTIR-ATR spectroscopy technology with chemometric analysis to construct a database for assessing honey authenticity. By employing partial least squares (PLS) models, effective differentiation between pure and adulterated honey was successfully achieved. All pure honey samples and standard honey samples (SBH) met the standards for non-adulteration. During the validation phase, 78 samples (including 6 pure honey samples and 72 adulterated honey samples) were accurately classified. The physicochemical properties of pure and adulterated honey met the requirements of EU regulations. Notably, this method can accurately identify and distinguish pure honey from adulterated honey across multiple dimensions, including solid solubility (SSD), moisture content, pH, free acidity, and sugar content. In summary, the combined use of FTIR-ATR spectroscopy technology and PLS models can serve as an efficient and non-destructive means of monitoring honey quality. This technology can detect any form of honey adulteration and establish recognition models for specific adulterants, further validating its effectiveness through validation sets. The research results indicate that the combination of FTIR-ATR spectroscopy and PLS regression analysis has significant advantages for rapid and accurate detection of honey adulteration. Additionally, accurately determining the degree of honey adulteration is crucial for establishing reasonable purchase and sale prices and serves as an important basis for addressing legal disputes arising from honey adulteration. This study provides a theoretical foundation for promoting this analytical method to the EU and other major honey production and trade regions worldwide. Future work recommendations include considering the plant sources of honey and their lower acidity and sugar concentrations when constructing the database to improve model sensitivity; given the diverse methods of adulteration in different countries and regions, practical case studies should be conducted to better guide the model’s future practical applications; and further analysis should be conducted on other types of adulterants, such as rapeseed honey and commercial adulterants.
https://doi.org/10.1016/j.foodcont.2024.110996
This public account article is for academic exchange only, and “original” indicates original translation; it does not represent this platform’s copyright claims. If there is any infringement, please contact the backend to modify or delete.
This study proposes a method combining FTIR-ATR spectroscopy technology with chemometric analysis to construct a database for assessing honey authenticity. By employing partial least squares (PLS) models, effective differentiation between pure and adulterated honey was successfully achieved. All pure honey samples and standard honey samples (SBH) met the standards for non-adulteration. During the validation phase, 78 samples (including 6 pure honey samples and 72 adulterated honey samples) were accurately classified. The physicochemical properties of pure and adulterated honey met the requirements of EU regulations. Notably, this method can accurately identify and distinguish pure honey from adulterated honey across multiple dimensions, including solid solubility (SSD), moisture content, pH, free acidity, and sugar content. In summary, the combined use of FTIR-ATR spectroscopy technology and PLS models can serve as an efficient and non-destructive means of monitoring honey quality. This technology can detect any form of honey adulteration and establish recognition models for specific adulterants, further validating its effectiveness through validation sets. The research results indicate that the combination of FTIR-ATR spectroscopy and PLS regression analysis has significant advantages for rapid and accurate detection of honey adulteration. Additionally, accurately determining the degree of honey adulteration is crucial for establishing reasonable purchase and sale prices and serves as an important basis for addressing legal disputes arising from honey adulteration. This study provides a theoretical foundation for promoting this analytical method to the EU and other major honey production and trade regions worldwide. Future work recommendations include considering the plant sources of honey and their lower acidity and sugar concentrations when constructing the database to improve model sensitivity; given the diverse methods of adulteration in different countries and regions, practical case studies should be conducted to better guide the model’s future practical applications; and further analysis should be conducted on other types of adulterants, such as rapeseed honey and commercial adulterants.
https://doi.org/10.1016/j.foodcont.2024.110996
This public account article is for academic exchange only, and “original” indicates original translation; it does not represent this platform’s copyright claims. If there is any infringement, please contact the backend to modify or delete.